Physical Chemistry of Polyelectrolytes: Surfactant Science 99

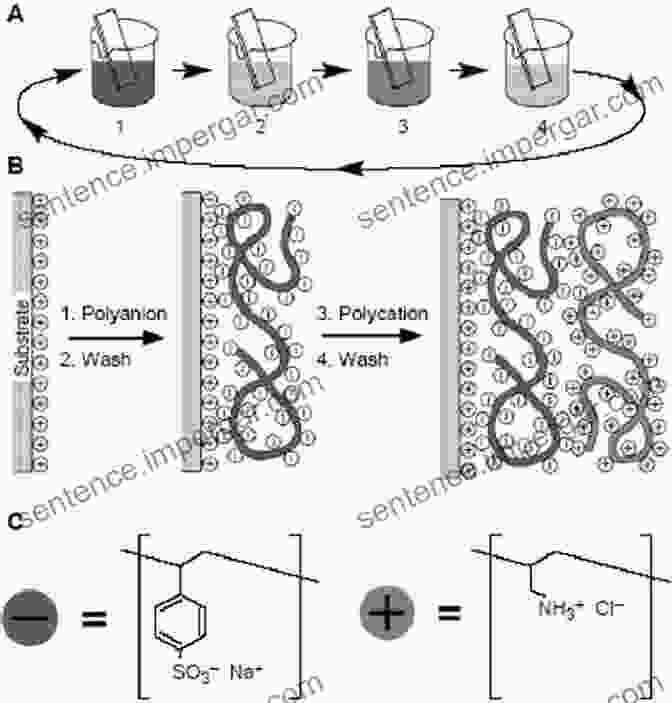
Polyelectrolytes are a fascinating class of materials that combine the properties of polymers and electrolytes. They are composed of long chains of repeating units, each of which contains an ionizable group. This unique structure gives polyelectrolytes a wide range of interesting properties, including the ability to interact with water, form micelles and emulsions, and exhibit electrokinetic phenomena.
5 out of 5
| Language | : | English |
| File size | : | 71996 KB |
| Screen Reader | : | Supported |
| Print length | : | 936 pages |
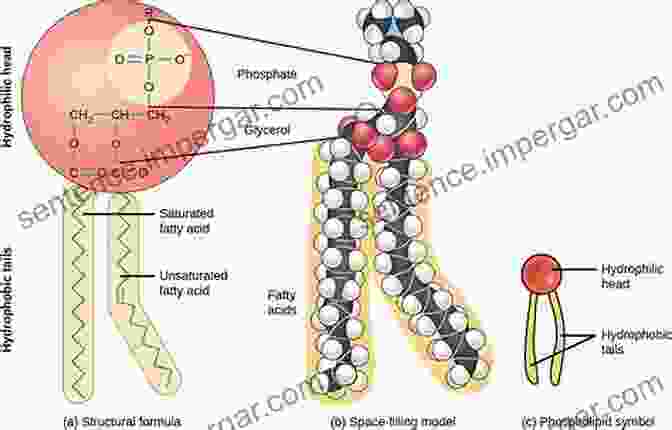
Surfactants are another important class of materials that are used in a wide variety of applications. They are typically composed of a hydrophilic head group and a hydrophobic tail group. This structure allows surfactants to interact with both water and oil, making them useful for applications such as detergency, emulsification, and foaming.
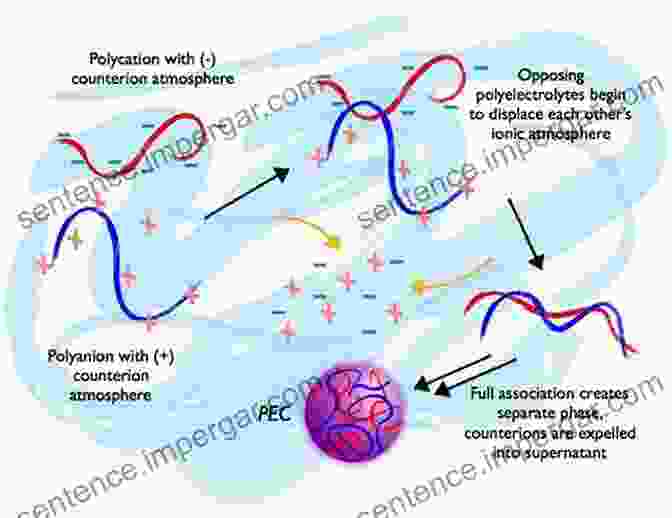
Polyelectrolyte surfactants are a combination of these two types of materials. They have a hydrophilic head group that is composed of a polyelectrolyte, and a hydrophobic tail group that is composed of a surfactant. This unique structure gives polyelectrolyte surfactants a wide range of interesting properties, including the ability to form complex structures, interact with both water and oil, and exhibit electrokinetic phenomena.
Physical Chemistry of Polyelectrolytes
The physical chemistry of polyelectrolytes is a complex and fascinating subject. Polyelectrolytes are polydisperse, meaning that they have a distribution of molecular weights. This distribution is important because it affects the properties of the polyelectrolyte. For example, the molecular weight of a polyelectrolyte affects its viscosity, elasticity, and solubility.
The solubility of polyelectrolytes is also affected by the pH of the solution. In acidic solutions, polyelectrolytes are typically more soluble than in basic solutions. This is because the protons in the acidic solution neutralize the negative charges on the polyelectrolyte, making it more soluble in water.
The physical chemistry of polyelectrolytes is also affected by the presence of other ions in the solution. Ions can interact with the polyelectrolyte, either by forming complexes with it or by screening its charges. This can affect the properties of the polyelectrolyte, such as its viscosity, elasticity, and solubility.
Applications of Polyelectrolyte Surfactants
Polyelectrolyte surfactants have a wide range of applications in both industrial and consumer products. They are used in detergents, shampoos, conditioners, paints, cosmetics, and food products. Polyelectrolyte surfactants are also used in a variety of industrial applications, such as oil and gas production, water treatment, and papermaking.

The unique properties of polyelectrolyte surfactants make them ideal for a variety of applications. They are able to interact with both water and oil, making them useful for applications such as detergency, emulsification, and foaming. Polyelectrolyte surfactants are also able to form complex structures, which makes them useful for applications such as drug delivery and gene therapy.
Polyelectrolyte surfactants are a fascinating and versatile class of materials with a wide range of applications. Their unique properties make them ideal for use in a variety of industrial and consumer products. As the field of physical chemistry continues to develop, we can expect to see even more applications for these amazing materials.
5 out of 5
| Language | : | English |
| File size | : | 71996 KB |
| Screen Reader | : | Supported |
| Print length | : | 936 pages |
Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
 Book
Book Novel
Novel Page
Page Chapter
Chapter Text
Text Story
Story Genre
Genre Reader
Reader Library
Library Paperback
Paperback E-book
E-book Magazine
Magazine Newspaper
Newspaper Paragraph
Paragraph Sentence
Sentence Bookmark
Bookmark Shelf
Shelf Glossary
Glossary Bibliography
Bibliography Foreword
Foreword Preface
Preface Synopsis
Synopsis Annotation
Annotation Footnote
Footnote Manuscript
Manuscript Scroll
Scroll Codex
Codex Tome
Tome Bestseller
Bestseller Classics
Classics Library card
Library card Narrative
Narrative Biography
Biography Autobiography
Autobiography Memoir
Memoir Reference
Reference Encyclopedia
Encyclopedia Jan Leja
Jan Leja James R Gillespie
James R Gillespie James F Simon
James F Simon James M Dennis
James M Dennis James E David
James E David Jayme Adelson Goldstein
Jayme Adelson Goldstein James C Kearney
James C Kearney James H Austin
James H Austin Janice J Beaty
Janice J Beaty James Scott Bell
James Scott Bell Jay Winik
Jay Winik Jason Fox
Jason Fox James Donaghy
James Donaghy James L Moody
James L Moody Janay Brun
Janay Brun Jamie Hunter
Jamie Hunter Janet Evans
Janet Evans James A Hessler
James A Hessler James Carl Nelson
James Carl Nelson Janie Mcgee
Janie Mcgee
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!

 Robert Louis StevensonSaratoga and the American Revolution: Pivotal Moments in American History
Robert Louis StevensonSaratoga and the American Revolution: Pivotal Moments in American History
 Ernest HemingwayWitness the Extraordinary Voyage of James Wood: A Captivating 18th-Century...
Ernest HemingwayWitness the Extraordinary Voyage of James Wood: A Captivating 18th-Century...
 Shannon SimmonsUnveiling the Timeless Wisdom of Vitruvius: A Journey into On Architecture...
Shannon SimmonsUnveiling the Timeless Wisdom of Vitruvius: A Journey into On Architecture... Rudyard KiplingFollow ·14.7k
Rudyard KiplingFollow ·14.7k Virginia WoolfFollow ·7.5k
Virginia WoolfFollow ·7.5k Thomas PynchonFollow ·8.5k
Thomas PynchonFollow ·8.5k Dan HendersonFollow ·18.3k
Dan HendersonFollow ·18.3k Billy PetersonFollow ·3.6k
Billy PetersonFollow ·3.6k Bernard PowellFollow ·16.8k
Bernard PowellFollow ·16.8k Derek CookFollow ·9.5k
Derek CookFollow ·9.5k Sam CarterFollow ·9.5k
Sam CarterFollow ·9.5k

 Jacob Foster
Jacob FosterPrinciples and Persons: The Legacy of Derek Parfit
Derek Parfit's 1984 book,...

 Leo Mitchell
Leo MitchellPartners For Life: Raise Support For Your Missionary Work...
Are you a missionary or ministry leader...

 Blake Kennedy
Blake KennedyOn Desperate Ground: A Gripping Account of World War II's...
Hampton Sides' "On...

 Duane Kelly
Duane KellyCriminal Minds Sociopaths Serial Killers And Other...
In the realm of criminology,...

 Craig Blair
Craig BlairHome Repair: The Ultimate Guide to Fix, Maintain, and...
Welcome to the...

 Elmer Powell
Elmer PowellThe Organic Grower Guide to Mycorrhizae Science for...
Unlock the Secrets of Soil...
5 out of 5
| Language | : | English |
| File size | : | 71996 KB |
| Screen Reader | : | Supported |
| Print length | : | 936 pages |